Read below to find out more:
RECOGNISING SIGNS AND SYMPTOMS OF SMA IN ADULTS
Have you experienced muscle weakness? Less severe forms of SMA can emerge and be diagnosed in adulthood.1,2 Compared to childhood SMA, adult SMA may have milder symptoms, but is still progressive. Compared to SMA in childhood, the course of SMA in adults can be more insidious and difficult to recognise.3
RECOGNISING SIGNS AND SYMPTOMS OF SMA IN ADULTS
Have you experienced muscle weakness? Less severe forms of SMA can emerge and be diagnosed in adulthood.1,2 Compared to childhood SMA, adult SMA may have milder symptoms, but is still progressive. Compared to SMA in childhood, the course of SMA in adults can be more insidious and difficult to recognise.3
This is why it is crucial to know how to identify the main signs and symptoms: if you are in doubt, ask your doctor.
In the image below, you can explore the signs and symptoms that are typically appearing in people living with SMA.
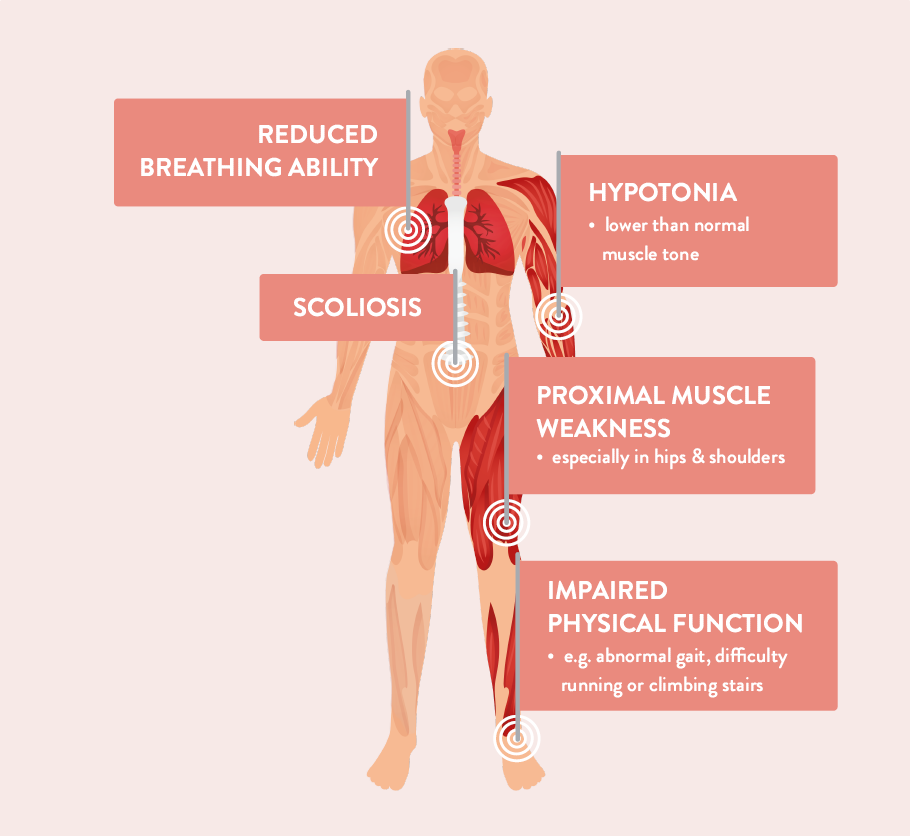
BREATHING ABILITY
- e.g. difficulty in raising the arms above the head
- curving of the spine
- especially in hips & shoulders
- e.g. difficulty running or climbing stairs
Hover on hotspots
to learn more
This is the checklist of the signs and symptoms organised around the domain they affect:
CARDINAL MOTOR SYMPTOMS1,4
- Symmetrical muscle weakness, usually proximal, especially of hip and shoulders
- Muscle wasting and muscle cramps
- Functional impairment, including: inability to run, gait alteration, difficulty getting up from the ground and climbing stairs
- Difficulty raising arms above head
- Hand tremors
- Tongue fasciculations (muscle twitches)
BONE SYMPTOMS1,4
- Skeletal deformities (scoliosis and hip dislocation)
- Joint contractures
RESPIRATORY SYMPTOMS1,5
- Reduced breathing ability
GENERAL SYMPTOMS1,5
- Fatigue
- Episodic weakness
A PATIENT’S PERSPECTIVE:
“I’m Normal. Just Different”
SAMUEL - A young adult living with SMA, who was diagnosed as an adolescent and his perspective on life with SMA.


IT’S IMPORTANT TO GET YOUR CONDITION DIAGNOSED BY A GENETIC TEST6
If you have already had a test, speak to your doctor to ensure you are receiving the most appropriate care.
In order to confirm SMA, a specialist neurologist requests a blood test that looks for specific alterations in your DNA – this is also referred to as an SMA gene‑deletion test.3,7,8
IT’S IMPORTANT TO GET YOUR CONDITION DIAGNOSED BY A GENETIC TEST6
If you have already had a test, speak to your doctor to ensure you are receiving the most appropriate care.
In order to confirm SMA, a specialist neurologist requests a blood test that looks for specific alterations in your DNA – this is also referred to as an SMA gene‑deletion test.3,7,8
THE IMPORTANCE OF HAVING A GENETIC TEST
Delays in diagnosis are common, which can potentially impact your care options9
You may be eligible for care options that are suited to a specific genetic diagnosis
You may have a condition that is not SMA but has similar characteristics (e.g. multifocal motor neuropathy), which may require different treatment10


A LOT HAS CHANGED IN THE QUALITY OF CARE FOR PEOPLE LIVING WITH SMA
People living with SMA can expect better outcomes through better care options. Improved management of the disease coupled with different care options could lead to a better quality of life for people living with SMA.6,11
Ongoing research is providing an ever-increasing understanding of the science behind SMA and care options.6,7,11
A LOT HAS CHANGED IN THE QUALITY OF CARE FOR PEOPLE LIVING WITH SMA
People living with SMA can expect better outcomes through better care options. Improved management of the disease coupled with different care options could lead to a better quality of life for people living with SMA.6,11
Ongoing research is providing an ever-increasing understanding of the science behind SMA and care options.6,7,11
SMA STANDARDS OF CARE HAVE BEEN UPDATED
Since the first Standards of Care publication in 2007, increasing evidence has shown that people living with SMA can expect better outcomes than ever before.
The 2017 update includes changes to the management and care you can expect to receive, helping you, your caregivers and healthcare professionals understand the different aspects of supportive care for SMA.7
You are placed at the very centre of care.
Summary of the latest SMA Standards of Care for SITTERS
Summary of the latest SMA Standards of Care for WALKERS
Treatment expectations for adults living with SMA have changed. Download the step-by-step guide below to gain tips about discussing your potential care options.


DISEASE STABILISATION REPRESENT MAJOR PROGRESS
Without intervention to stabilise the disease, people living with type II/III spinal muscular atrophy (SMA) will see a progression of their disease over time.15,16
Your perspective and experiences with SMA are critically important in the development of new therapies. In 2015, a European survey was completed by people living with type II/III SMA and by caregivers. The results showed that:15
DISEASE STABILISATION REPRESENT MAJOR PROGRESS
Without intervention to stabilise the disease, people living with type II/III spinal muscular atrophy (SMA) will see a progression of their disease over time.15,16
Your perspective and experiences with SMA are critically important in the development of new therapies. In 2015, a European survey was completed by people living with type II/III SMA and by caregivers. The results showed that:15
felt that a new care option that would stabilise their disease course would represent MAJOR PROGRESS
See our summary on what other people affected by SMA think about the disease:
Things that people often take for granted like being able to shower or bathe, using the toilet, and being able to eat independently are essential for everybody but especially for those people living with SMA, because it means sustaining independence and dignity.15
VOICES FROM PEOPLE LIVING WITH SMA
Once thought to be primarily a childhood disease, SMA can be diagnosed in adolescents and adults.18 Meet Michel and Samuel and put yourself in the shoes of adults living with SMA or their family.
A CAREGIVER’S PERSPECTIVE:
“I wish I had pushed harder”
MICHEL - A caregiver of a young adult living with SMA and his family’s journey.


Now is the time to find out how the latest changes to SMA management could potentially benefit you.
Ask your doctor for a referral to an SMA specialist
About Together in SMA
At Biogen, we are committed to supporting those with spinal muscular atrophy and their care team. Our hope for Together in SMA is that, by providing information, we can help you to get the most up-to-date care, and to help guide conversations with your doctors.
Here you'll find information about spinal muscular atrophy and its symptoms, insights into care options, and perspectives from people living with SMA, experienced caregivers and healthcare professionals on topics ranging from nutrition to adaptive equipment.
References
1. Montes J. Spinal Muscular Atrophy in Adults. [online] [cited 2020 Nov 30]. Available from: URL: http://www.neuropt.org/docs/degenerative-diseases-sig/spinalmuscular-atrophy-in-adults.pdf?sfvrsn=8d2aae96_2.
2. Juntas Morales R, Pageot N, Taieb G, Camu W. Adult-onset spinal muscular atrophy: and update. Rev Neurol (Paris) 2017;173(5):308-319.
3. Wang CH, et al. Consensus statement for standard of care in spinal muscular atrophy. J Child Neurol 2007;22(8):1027-1049.
4. Arnold WD, et al. Muscle Nerve. 2015;51(2):157-67.
5. Wijngaarde CA, et al. Orphanet J Rare Dis. 2020;15(1):88.
6. Mercuri E, et al. Diagnosis and management of spinal muscular atrophy: Part 1: Recommendations for diagnosis, rehabilitation, orthopedic and nutritional care. Neuromuscl Disord 2018;28(2):103-115.
7. Bharucha-Goebel D, Kaufmann P. Treatment Advances in Spinal Muscular Atrophy. Curr Neurol Neurosci Rep 2017;17(11):91.
8. Prior TW, Finanger E, Leach ME. Spinal muscular atrophy. NCBI Bookshelf Website. [online] 2019 Nov 14 [cited 2010 Aug 24]. Available from: URL: https://www.ncbi.nlm.nih.gov/books/NBK1352/.
9. Lin CW, Kalb SJ, Yeh WS. Delay in Diagnosis of Spinal Muscular Atrophy: A Systematic Literature Review. Pediatr Neurol 2015;53:293-300.
10. Visser J, et al. Mimic syndromes in sporadic cases of progressive spinal muscular atrophy. Neurology 2002;58(11):1593-1596.
11. Finkel R, et al. Diagnosis and management of spinal muscular atrophy: Part 2: Pulmonary and acute care; medications, supplements and immunizations; other organ systems; and ethics. Neuromuscul Disord 2018;28(3):197-207.
12. National Center for Biotechnology Information. Spinal muscular atrophy. Pubmed website. [online] [cited 2021 Mar 22] Available from: URL: https://pubmed.ncbi.nlm.nih.gov/?term=%28spinal+&muscular+&atrophy%29+&AND+%28%&28%222018&%2F01%2F01&%22%5BDate&+- +Publication&%5D+%3A+&%222018%&2F12%2F31%22&%5BDate+-+&Publication&%5D%29%29&sort=.
13. Wang CH et al. National Center for Biotechnology Information. Spinal muscular atrophy. PubMed website. [online] [cited 2021 Mar 22] Available from: URL: https://pubmed.ncbi&.nlm.nih.gov/?term=%&28spinal+muscular&+atrophy%29&+AND+%28%28%222010&%2F01%2F01%22&%5BDate+- +Publication&%5D+%3A+%222010%&2F12%2F31%22%&5BDate+-+Publication&%5D%29%29&sort=.
14. Search for ‘Recruiting, Active, not recruiting. Enrolling by invitation Studies. Spinal Muscular Atrophy. ClinicalTrials.gov. [online] [cited 2021 Mar 22] Available from: URL: https://www.clinicaltrials.gov/ct2/results?cond=%22Spinal+muscular+atrophy%22&term=&type=&rslt=&recrs=b&recrs=a&recrs=f&recrs=d&age_v=&gndr=&intr=&titles=&outc=&spons=&lead=&id=&cntry=&state=&city=&dist=&locn=&rsub=&strd_s=&strd_e=&prcd_s=&prcd_e=&sfpd_s=&sfpd_e=&rfpd_s=&rfpd_e=&lupd_s=&lupd_e=&sort=.
15. Rouault F, et al. Disease impact on general well-being and therapeutic expectations of European Type II and Type III spinal muscular atrophy patients. Neuromuscul Disord 2017;27(5):428-438.
16. Kaufmann P, et al. Prospective cohort study of spinal muscular atrophy types 2 and 3. Neurol 2012;79:1889-1897.
17. Wan H et al. “Getting ready for the adult world”: how adults with spinal muscular atrophy perceive and experience healthcare, transition and well-being. Orphanet J Rare Dis. 2019;14(1):74.
18. NIH. Spinal Muscular Atrophy Factsheet: https://www.ninds.nih.gov/Disorders/Patient-Caregiver-Education/Fact-Sheets/Spinal-Muscular-Atrophy-Fact-Sheet.







































